Demand for 1,8-Dinitroanthraquinone in USA | Global Market Analysis Report

Demand for 1,8 -Dinitroanthraquinone in USA 2025 to 2035
The demand for 1,8-dinitroanthraquinone in the USA is forecast to grow from USD 1.5 million in 2025 to approximately USD 2.0 million by 2035, recording an absolute increase of USD 0.5 million over the forecast period. This translates into total growth of 33.3%, with demand forecast to expand at a compound annual growth rate (CAGR) of 2.9% between 2025 and 2035. Expansion is supported by wider usage of high-grade intermediates required for synthesis efficiency, pharmaceutical formulation accuracy, and chemical processing reliability across major research and production corridors. Purity ≥98% holds a 64.8% share in 2025, favored for compatibility with existing equipment and its stable performance profile across pharmaceutical workflows.
Purity ≥99% gains interest among facilities seeking enhanced molecular precision for advanced synthesis. Pharmaceutical manufacturing accounts for 52.7% of demand due to its reliance on controlled-purity compounds that support yield stability, process safety, and predictable synthesis pathways. Dye production and broader chemical processing account for the remaining share as specialty compound needs evolve across regional manufacturing clusters. The Northeast leads with a 3.2% CAGR, supported by concentrated research facilities and a strong pharmaceutical presence, while the West follows at 3.0% through biotechnology density and chemical capabilities. The South at 2.8% and the Midwest at 2.6% sustain adoption by expanding chemical sites and modernizing regional pharmaceutical operations.
Quick Stats for USA 1,8-Dinitroanthraquinone Industry
- USA 1,8-Dinitroanthraquinone Sales Value (2025): USD 1.5 million
- USA 1,8-Dinitroanthraquinone Forecast Value (2035): USD 2.0 million
- USA 1,8-Dinitroanthraquinone Forecast CAGR: 2.9%
- Leading Purity Category in USA 1,8-Dinitroanthraquinone Demand: Purity ≥98% (64.8%)
- Key Growth Regions in USA 1,8-Dinitroanthraquinone Demand: Northeast, West, South, and Midwest
- Regional Leadership: Northeast holds the leading position in demand
- Key Players in USA 1,8-Dinitroanthraquinone Demand: Qingdao Haiwan Specialty Chemicals, Zhejiang Tesin Chemical, Hubei Qifei Pharmaceutical Chemical
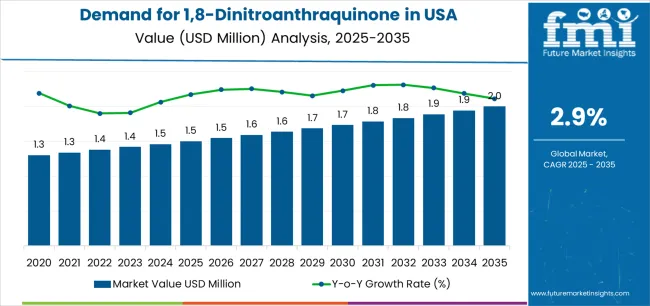
From 2030 to 2035, demand is forecast to grow from USD 1.7 million to USD 2.0 million, adding another USD 0.3 million, which constitutes 60.0% of the overall ten-year expansion. This period is expected to be characterized by expansion of pharmaceutical synthesis technologies, integration of specialized chemical processing systems and quality management networks, and development of advanced intermediate pathways across different pharmaceutical applications. The growing adoption of precision synthesis principles and enhanced quality control requirements, particularly in Northeast and West regions, will drive demand for more sophisticated chemical systems and specialized purity specifications.
Between 2020 and 2025, USA 1,8-dinitroanthraquinone demand experienced steady expansion, driven by increasing pharmaceutical operational requirements in specialty chemical sectors and growing awareness of high-purity compound benefits for synthesis enhancement and operational reliability. The sector developed as pharmaceutical manufacturers and chemical companies, especially in major research corridors, recognized the need for specialized intermediates and reliable quality management to achieve synthesis targets while meeting pharmaceutical expectations and efficiency requirements. Chemical suppliers and pharmaceutical manufacturers began emphasizing proper performance optimization and application integration to maintain synthesis effectiveness and commercial viability.
USA 1,8 -Dinitroanthraquinone Key Takeaways
| Metric | Value |
|---|---|
| USA 1,8-Dinitroanthraquinone Sales Value (2025) | USD 1.5 million |
| USA 1,8-Dinitroanthraquinone Forecast Value (2035) | USD 2.0 million |
| USA 1,8-Dinitroanthraquinone Forecast CAGR (2025-2035) | 2.9% |
Why is the USA 1,8-Dinitroanthraquinone Demand Growing?
Demand expansion is being supported by the accelerating emphasis on pharmaceutical manufacturing excellence and chemical processing optimization nationwide, with the USA maintaining its position as a pharmaceutical and chemical synthesis innovation leadership region, and the corresponding need for effective high-purity intermediate systems for drug development, synthesis efficiency, and quality assurance integration. Modern pharmaceutical strategies rely on 1,8-dinitroanthraquinone technologies to ensure synthesis competitiveness, quality compliance, and optimal pathway achievement toward efficiency-focused pharmaceutical operations. Chemical operational requirements necessitate comprehensive intermediate solutions including advanced purity processing, molecular management capabilities, and performance control infrastructure to address diverse pharmaceutical needs and synthesis specifications.
The growing emphasis on pharmaceutical quality standards and increasing federal and state-level chemical regulations, particularly synthesis excellence commitments across the USA, are driving demand for high-purity intermediate systems from proven chemical suppliers with appropriate pharmaceutical expertise and quality management capabilities. Pharmaceutical manufacturers and chemical companies are increasingly investing in specialized compound sourcing and integrated synthesis solutions to enhance pharmaceutical profiles, access efficiency trends, and demonstrate chemical leadership in competitive pharmaceutical environments. Chemical policies and quality control requirements are establishing standardized intermediate pathways that require high-purity systems and performance assurance, with USA pharmaceutical operations often pioneering large-scale implementation of specialized chemical technologies.
How Is the USA 1,8-Dinitroanthraquinone Market Segmented by Purity, Application, and Region?
Demand is segmented by purity grade, application, and region. By purity grade, sales are divided into purity ≥98%, purity ≥99%, and other purity configurations. In terms of application, sales are segmented into pharmaceutical manufacturing, dye production, chemical processing, and others. Regionally, demand is divided into Northeast, West, South, and Midwest, with Northeast representing a key growth and innovation hub for 1,8-dinitroanthraquinone technologies.
By Purity Grade, Purity ≥98% Segment Accounts for 64.8% Share
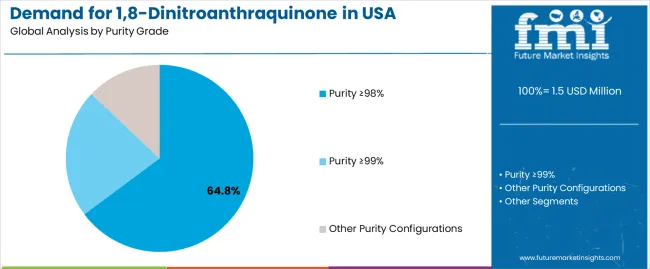
The purity ≥98% segment is projected to account for 64.8% of USA 1,8-dinitroanthraquinone demand in 2025, making it the leading purity category across the sector. This dominance reflects the operational balance and quality suitability of high-grade chemical systems for existing pharmaceutical facilities and chemical applications where synthesis performance is optimized through controlled purity specifications. In the USA, where substantial pharmaceutical infrastructure requires intermediate integration without complete equipment replacement, purity ≥98% systems provide practical pathways for synthesis enhancement while maintaining operational efficiency continuity. Continuous innovations are improving chemical effectiveness, molecular stability characteristics, and application compatibility parameters, enabling manufacturers to achieve high performance standards while minimizing operational cost increases. The segment’s strong position is reinforced by the extensive existing pharmaceutical infrastructure requiring intermediate adoption and growing availability of purity ≥98% technology suppliers with proven commercial experience.
- Processing compatibility and existing equipment integration make purity ≥98% systems the preferred technology for enhancing operating pharmaceutical facilities and chemical installations.
- Technical maturity and commercial demonstration track records are enhancing operator confidence and project viability across large-scale deployment initiatives.
By Application, Pharmaceutical Manufacturing Segment Accounts for 52.7% Share
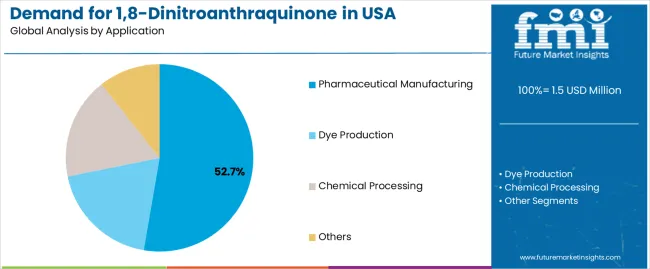
Pharmaceutical manufacturing applications are expected to represent 52.7% of USA 1,8-dinitroanthraquinone demand in 2025, highlighting the critical importance of drug development requiring specialized chemical solutions. Pharmaceutical facilities including research laboratories, production operations, specialty synthesis, and pharmaceutical manufacturing applications generate consistent demand for chemical systems that are technically and economically favorable for high-purity applications. The segment benefits from synthesis characteristics that often provide superior operational reliability compared to standard alternatives, reducing complexity and costs. Pharmaceutical applications also access enhanced efficiency through specialized compounds that improve synthesis performance and appeal. In the USA, where pharmaceutical innovation represents substantial portions of chemical development, specialized compound deployment requires 1,8-dinitroanthraquinone integration across diverse pharmaceutical operations. In Northeast and West regions, where pharmaceutical concentrations are significant, 1,8-dinitroanthraquinone demand is elevated by emphasis on maintaining synthesis efficiency while achieving pharmaceutical integration targets.
- Chemical concentration and favorable synthesis economics make this the largest application segment for 1,8-dinitroanthraquinone technologies.
- Quality preferences and synthesis requirements drive consistent demand across research laboratories, production operations, specialty synthesis, and pharmaceutical manufacturing applications.
What are the Drivers, Restraints, and Key Trends in the USA 1,8-Dinitroanthraquinone Demand?
USA 1,8-dinitroanthraquinone demand is advancing steadily due to increasing pharmaceutical efficiency and growing recognition of specialized chemical necessity for pharmaceutical development, with Northeast region serving as a key driver of innovation and application development. The sector faces challenges including competition from alternative compounds, purity cost considerations, and ongoing concerns regarding initial investment costs and supply chain requirements. Federal pharmaceutical guidelines and state-level quality initiatives, particularly chemical programs in Northeast and West regions, continue to influence compound selection and deployment timelines.
Expansion of Pharmaceutical Quality Requirements and Synthesis Standards
The enhancement of pharmaceutical quality regulations, gaining particular significance through drug development guidelines and synthesis excellence campaigns, is enabling chemical suppliers to achieve differentiation without prohibitive investment costs, providing predictable demand patterns through pharmaceutical requirements and synthesis efficiency preferences. Enhanced quality standards offering substantial opportunities for high-purity chemical systems and pharmaceutical applications provide foundational dynamics while allowing suppliers to secure pharmaceutical agreements and synthesis partnerships. These trends are particularly valuable for first-mover suppliers and premium chemical development that require substantial purity investments without immediate cost advantages.
Integration of Advanced Chemical Technologies and Quality Monitoring Systems
Modern chemical suppliers and manufacturers are establishing advanced quality management networks and centralized synthesis monitoring facilities that improve operational efficiency through purity standardization and economies of scale. Integration of specialized chemical systems, real-time quality monitoring, and coordinated synthesis management enables more efficient compound operation across multiple pharmaceutical sources. Advanced chemical concepts also support next-generation pharmaceutical applications including specialized facility integration, synthesis cluster optimization, and regional chemical supply networks that optimize system-level economics while enabling comprehensive efficiency across pharmaceutical regions, with USA developments increasingly adopting collaborative chemical models to reduce individual operator costs and accelerate deployment.
How Does USA 1,8-Dinitroanthraquinone Demand Vary by Key Region?
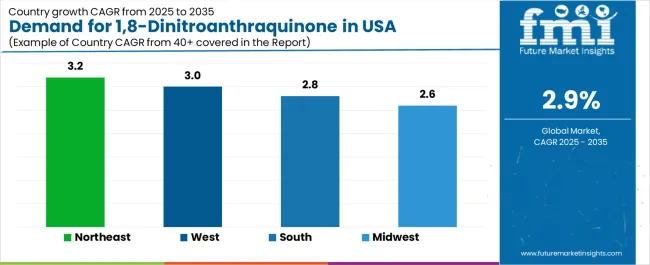
| Region | CAGR (2025-2035) |
|---|---|
| Northeast | 3.2% |
| West | 3.0% |
| South | 2.8% |
| Midwest | 2.6% |
The USA 1,8-dinitroanthraquinone demand is witnessing consistent growth, supported by rising pharmaceutical efficiency, expanding quality requirements, and the deployment of advanced chemical technologies across regions. Northeast leads the nation with a 3.2% CAGR, reflecting progressive pharmaceutical trends, substantial research innovation, and early adoption of premium chemical systems. West follows with a 3.0% CAGR, driven by extensive pharmaceutical infrastructure, favorable research demographics, and concentration of chemical operations that enhance application development. South grows at 2.8%, as pharmaceutical modernization and synthesis efficiency opportunities increasingly drive compound deployment. Midwest demonstrates growth at 2.6%, supported by expanding chemical facilities and regional pharmaceutical initiatives.
Why Does the Northeast Lead USA 1,8-Dinitroanthraquinone Growth with a 3.2% CAGR?
Demand for 1,8-dinitroanthraquinone in Northeast is projected to exhibit exceptional growth with a CAGR of 3.2% through 2035, driven by progressive pharmaceutical efficiency preferences, substantial research development creating premium chemical opportunities, and concentration of innovation across Massachusetts and surrounding states. As the dominant region with extensive pharmaceutical infrastructure and efficiency-focused operational policies, Northeast’s emphasis on comprehensive drug development and chemical leadership is creating significant demand for advanced 1,8-dinitroanthraquinone systems with proven performance and reliable application potential. Major pharmaceutical manufacturers and chemical suppliers are establishing comprehensive compound development programs to support research innovation and premium chemical deployment across diverse applications.
- Pharmaceutical efficiency trends and synthesis cost preferences are requiring comprehensive quality strategies and chemical solutions, driving demand for 1,8-dinitroanthraquinone systems with demonstrated pharmaceutical capabilities and permanent performance assurance throughout diverse synthesis operations.
- Innovation ecosystem strength and investment capital availability are supporting deployment of next-generation chemical technologies and novel application pathways that enhance commercial viability, reduce synthesis costs, and create new pharmaceutical opportunities across research and chemical applications, positioning Northeast as a national chemical leadership region.
How Is the West Leveraging Its Pharmaceutical Infrastructure to Strengthen Demand?
Demand for 1,8-dinitroanthraquinone in West is expanding at a CAGR of 3.0%, supported by extensive pharmaceutical facilities including biotechnology companies, research operations, and chemical establishments generating concentrated demand favorable for high-purity chemical systems. The region’s operational characteristics, featuring substantial pharmaceutical operations and synthesis efficiency requirements ideal for specialized integration, provide natural advantages. Pharmaceutical industry expertise concentrated in California, Washington, and regional research corridors facilitates application development and operational management. Chemical suppliers and manufacturers are implementing comprehensive compound strategies to serve expanding efficiency-focused requirements throughout West.
- Pharmaceutical concentration and favorable application economics are creating opportunities for specialized chemical suppliers that can integrate compound systems with existing pharmaceutical operations.
- Synthesis efficiency positioning and pharmaceutical awareness are building regional competitive advantages in chemical applications, enabling comprehensive pharmaceutical development and research cluster enhancement that meets synthesis targets while accessing efficiency pricing opportunities.
Why Is the South Sustaining Steady Growth Through Chemical Management Efficiency?
Demand for 1,8-dinitroanthraquinone in South is growing at a CAGR of 2.8%, driven by substantial chemical management facilities from pharmaceutical operations, manufacturing services, and regional processing requiring high-purity compound pathways. The region’s pharmaceutical base, supporting critical chemical operations, is increasingly adopting compound technologies to maintain competitiveness while meeting efficiency expectations. Manufacturers and chemical suppliers are investing in compound integration systems and regional supply infrastructure to address growing quality requirements.
- Chemical management modernization imperatives and operational competitiveness concerns are facilitating adoption of 1,8-dinitroanthraquinone technologies that enable continued operations while achieving efficiency enhancement across pharmaceutical, manufacturing, and chemical processing facilities.
- Pharmaceutical efficiency opportunities including regional chemical development and compound utilization for enhanced commercial operations are creating unique regional advantages and diversified application types throughout South pharmaceutical operations.
How Is the Midwest Expanding 1,8-Dinitroanthraquinone Adoption Through Facility Development?
Demand for 1,8-dinitroanthraquinone in Midwest is advancing at a CAGR of 2.6%, supported by expanding chemical facilities, regional pharmaceutical development including specialized processing applications, and growing emphasis on compound solutions across the region. Chemical modernization and pharmaceutical facility expansion are driving consideration of high-purity compounds as operational enhancement pathways. Pharmaceutical companies and chemical suppliers are developing regional capabilities to support emerging compound deployment requirements.
- Chemical expansion and operational diversification are creating economic drivers for quality technologies and 1,8-dinitroanthraquinone deployment across pharmaceutical and chemical facilities seeking competitive differentiation pathways.
- Regional pharmaceutical cooperation and coordinated chemical development are establishing consistent compound environments and shared operational infrastructure that support multi-state pharmaceutical projects throughout Midwest chemical operations.
What Defines the Competitive Landscape of USA 1,8-Dinitroanthraquinone Demand?
Demand for 1,8-dinitroanthraquinone in the United States is defined by competition among specialized chemical manufacturers, pharmaceutical companies, and synthesis solution providers, with major chemical corporations maintaining significant influence through supply chain resources and technology development capabilities. Companies are investing in purity technology advancement, molecular optimization, distribution network structures, and comprehensive application services to deliver effective, reliable, and scalable chemical solutions across the USA pharmaceutical and chemical applications. Strategic partnerships, technology infrastructure development, and first-mover application execution are central to strengthening competitive positioning and presence across pharmaceutical applications, research facilities, and synthesis applications.
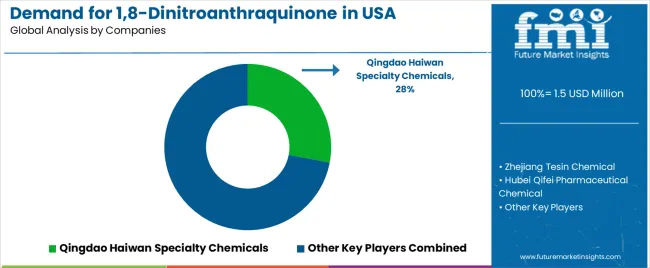
Qingdao Haiwan Specialty Chemicals, an internationally recognized chemical leader, leads with a 28% share, offering a comprehensive supply of 1,8-dinitroanthraquinone, including manufacturing, technology, and distribution services, with a focus on pharmaceutical applications, performance reliability, and cost optimization across USA operations. Zhejiang Tesin Chemical, operating nationally with extensive USA distribution, provides integrated chemical solutions leveraging engineering expertise, quality assurance development, and large-scale manufacturing capabilities.
Hubei Qifei Pharmaceutical Chemical delivers full-service 1,8-dinitroanthraquinone processing, including synthesis technology, performance testing, and supply management, serving USA and international pharmaceutical projects. Shengao Chemical Industry emphasizes comprehensive specialty chemical solutions with integrated technology, quality control, and distribution capabilities, leveraging pharmaceutical sector expertise. Jiangsu Yabang Dyestuff provides 1,8-dinitroanthraquinone application development and quality assurance services for pharmaceutical and chemical applications across the USA.
USA 1,8-Dinitroanthraquinone Demand – Stakeholder Contribution Framework
1,8-Dinitroanthraquinone represents critical chemical infrastructure for enhancing pharmaceutical productivity, supporting synthesis efficiency, and enabling quality applications essential for achieving pharmaceutical performance targets. With the demand projected to reach USD 2.0 million by 2035, driven by pharmaceutical efficiency, quality requirements, and technology advancement, the sector stands at the intersection of pharmaceutical innovation, synthesis excellence, and chemical development. The compound ecosystem spanning synthesis systems, supply chain networks, quality monitoring facilities, and application development infrastructure requires coordinated action across chemical suppliers, manufacturers, pharmaceutical distributors, regulatory authorities, research institutions, and pharmaceutical organizations to unlock its full value potential while addressing the technical complexities of large-scale chemical management and consistent performance delivery.
How Governments Could Accelerate Development and Performance Standards?
- Enhanced Chemical Safety Guidelines and Pharmaceutical Standards: Expand and clarify FDA pharmaceutical regulations by increasing performance specifications, simplifying certification procedures, and ensuring long-term policy consistency to improve dynamics and reduce regulatory uncertainty for 1,8-dinitroanthraquinone deployment.
- Research and Development Funding: Provide federal funding for chemical technology research, advanced synthesis system development, and performance enhancement methods through grants and public-private partnerships that advance technical capabilities while reducing operational costs.
- Pharmaceutical Infrastructure Investment: Support development of chemical supply networks, quality testing facility establishment, and pharmaceutical infrastructure that reduces operational costs while building shared compound capacity accessible to multiple pharmaceutical applications.
- Chemical Equipment Clarity: Establish clear frameworks for compound labeling, performance claim verification, and quality content standards that address operator information needs while ensuring accurate equipment representation.
- Equipment Quality Standards: Define standardized performance specifications, testing protocols, and certification requirements for 1,8-dinitroanthraquinone products that ensure safety and effectiveness while enabling domestic supply comparison and technology evaluation.
- Pharmaceutical Research Support: Support development of domestic pharmaceutical compound research, synthesis optimization studies, and supply chain development through federal pharmaceutical programs that enable regional compound development.
How Industry Bodies Could Strengthen Sector Development?
- Performance Standards and Chemical Best Practices: Define standardized compound specifications, quality metrics, and testing protocols for 1,8-dinitroanthraquinone systems and applications that ensure consistency and effectiveness while enabling product comparison and technology evaluation.
- Supply Chain Assessment Methodologies: Develop comprehensive evaluation frameworks for assessing compound quality, operational capacity, and supply reliability that reduce sourcing uncertainty while building supplier confidence.
- Technical Training and Certification: Establish certification programs, compound training curricula, and professional development pathways for chemical technicians, manufacturers, and performance specialists that ensure adequate skilled workforce for sector expansion.
- Application Development Frameworks: Create standardized methodologies for measuring compound effectiveness accounting for purity, synthesis compatibility, and operational performance that enable credible product benefit verification.
- Pharmaceutical Education and Acceptance Programs: Coordinate pharmaceutical outreach, educational initiatives, and transparent communication to address 1,8-dinitroanthraquinone benefits, installation safety, and operational advantages, building acceptance for compound development.
- Supply Chain Coordination Initiatives: Facilitate coordination among manufacturers, distributors, and pharmaceutical operators, addressing bottlenecks, capacity constraints, and cost-reduction opportunities across 1,8-dinitroanthraquinone supply chains.
How Chemical Suppliers Could Capture Value and Drive Innovation?
- Advanced Chemical Technology Development: Invest in enhanced synthesis systems, molecular processes, and performance monitoring technologies that improve operational efficiency, reduce quality costs, and enhance compound consistency beyond conventional chemical methods.
- Standardized Product Development: Develop pre-qualified compound specifications with standardized performance parameters that reduce installation costs, accelerate deployment timelines, and enable consistent operation with quality assurance advantages.
- Application Support and Technical Services: Incorporate installation assistance, performance testing, and technical optimization services that maximize compound performance while minimizing application complexity and operational costs throughout product lifecycles.
- Supply Chain Integration: Pioneer comprehensive supply chain systems with improved sourcing reliability and enhanced cost structures that enable consistent compound availability and quality management beyond traditional supply approaches.
- Specialized Application Development: Create commercial pathways for 1,8-dinitroanthraquinone in specialized pharmaceutical categories, chemical applications, and research facilities that generate additional revenue streams while expanding applications.
- Comprehensive Service Offerings: Provide integrated solutions spanning compound supply, technical services, quality assurance, and application support that simplify adoption for manufacturers while creating recurring business opportunities.
How Pharmaceutical Manufacturers Could Optimize Chemical Enhancement Strategies?
- Comprehensive Chemical Assessment: Conduct detailed operational analysis identifying compound opportunities, equipment compatibility, and application suitability that inform strategic pharmaceutical development roadmaps and chemical selection.
- Installation Integration and Feasibility Studies: Evaluate compound options based on specific facility characteristics, operational capabilities, and performance requirements through rigorous technical analysis before major chemical commitments.
- Phased Implementation Roadmaps: Develop staged deployment plans beginning with pilot installations, progressing through operational testing, and scaling to full facility deployment that manage development risks while building operational experience.
- Supply Chain Partnership Development: Engage in long-term supplier relationships ensuring consistent compound quality and availability that reduce individual installation costs while enabling smaller operators to participate in high-purity chemical sectors.
- Operational Positioning and Cost Optimization: Secure operational efficiency positioning, develop premium pharmaceutical services, and access compound technology benefits that improve facility economics and enable differentiation.
- Worker Communication and Training: Build relationships with worker organizations, facility managers, and operational groups early in compound development that facilitate equipment acceptance while addressing operational questions and building chemical support.
How Research Companies Could Lead Chemical Integration?
- Chemical Product Portfolio Development: Leverage research expertise and existing contractor relationships to identify, integrate, and promote 1,8-dinitroanthraquinone systems in project designs and equipment specifications providing commercial chemical services to efficiency-conscious research operators.
- Quality Assurance Network Development: Develop comprehensive testing infrastructure connecting compound sourcing to finished project quality that enable regional chemical management while generating quality verification revenue.
- Integrated Project Development: Combine 1,8-dinitroanthraquinone integration, research capacity, and service operations into comprehensive project solutions that simplify compound development for operators while capturing value across the supply chain.
- Performance Benefit Communication: Utilize compound efficiency properties for research applications that generate operator education revenue while supporting project goals in finished research products.
- Specialized Research Applications: Pioneer targeted project configurations leveraging compound research and operator access that enable specific research applications and chemical generation.
- Technical Quality Management: Apply quality control, large project management, and safety compliance capabilities from research operations to 1,8-dinitroanthraquinone equipment development.
How Biotechnology Companies Could Unlock Pharmaceutical Innovation?
- Pharmaceutical Enhancement Development: Develop specialized biotechnology products incorporating 1,8-dinitroanthraquinone benefits through compound systems for efficiency-conscious manufacturers with chemical preferences.
- Quality Applications: Provide biotechnology compound solutions monetizing 1,8-dinitroanthraquinone purity and efficiency benefits for biotechnology developers lacking sufficient chemical options.
- Installation Integration Support: Create dedicated infrastructure targeting 1,8-dinitroanthraquinone biotechnology applications and quality assurance systems that generate stable operations through compound contracts while supporting multiple biotechnology applications.
- Premium Positioning: Issue dedicated product lines funding 1,8-dinitroanthraquinone biotechnology deployment that attract efficiency-focused manufacturers while supporting biotechnology compound infrastructure development.
- Quality Assurance Systems: Develop quality testing, performance verification, and manufacturer feedback systems addressing compound performance, purity delivery, and efficiency benefits that enable broader manufacturer participation in biotechnology compound sectors.
How Investors and Financial Enablers Could Unlock Growth?
- Chemical Infrastructure Investment: Provide growth capital for pioneering commercial-scale 1,8-dinitroanthraquinone manufacturing facilities demonstrating technology viability and establishing operational track records that enable subsequent financing.
- Supply Chain Development Funding: Finance shared compound sourcing and manufacturing infrastructure serving multiple pharmaceutical operators through infrastructure investments generating returns from supply contracts and chemical services.
- Technology Company Growth Capital: Support 1,8-dinitroanthraquinone technology developers, chemical equipment firms, and service providers scaling operations and expanding geographic presence through growth equity investments.
- Portfolio Investment: Acquire portfolios of application development projects from operating suppliers providing upfront capital to developers while capturing long-term value from expansion.
- Strategic Industry Consolidation: Finance acquisitions consolidating fragmented chemical suppliers, compound firms, and application developers creating integrated chemical platforms with comprehensive capabilities.
- Performance-Based Financing: Structure innovative financing arrangements linking capital costs and returns to measurable quality performance, supply reliability, and verified penetration that align stakeholder incentives around project success.
Key Players in USA 1,8-Dinitroanthraquinone Demand
- Qingdao Haiwan Specialty Chemicals
- Zhejiang Tesin Chemical
- Hubei Qifei Pharmaceutical Chemical
- Shengao Chemical Industry
- Jiangsu Yabang Dyestuff
- Henan Tianfu Chemical
- Changzhou Jingye Chemical
- Shanghai Dyestuff Research Institute
- Eastman Chemical Company
- Huntsman Corporation
What Is the Scope of the USA 1,8-Dinitroanthraquinone Report?
| Item | Value |
|---|---|
| Quantitative Units | USD 2.0 million |
| Purity Grade | Purity ≥98%, Purity ≥99%, Other Purity Configurations |
| Application | Pharmaceutical manufacturing, dye production, chemical processing, others |
| Regions Covered | Northeast, West, South, Midwest |
| Key Companies Profiled | Qingdao Haiwan Specialty Chemicals, Zhejiang Tesin Chemical, Hubei Qifei Pharmaceutical Chemical, Shengao Chemical Industry |
| Additional Attributes | Sales by purity grade and application segment, regional demand trends across Northeast, West, South, and Midwest, competitive landscape with established chemical suppliers and specialized pharmaceutical manufacturers, manufacturer preferences for purity ≥98% versus other chemical technologies, integration with pharmaceutical efficiency programs and quality policies particularly advanced in Northeast region, innovations in compound efficiency and performance enhancement technologies |
USA 1,8-Dinitroanthraquinone Demand by Segments
Purity Grade:
- Purity ≥98%
- Purity ≥99%
- Other Purity Configurations
Application:
- Pharmaceutical Manufacturing
- Dye Production
- Chemical Processing
- Others
Region:
- Northeast
- West
- South
- Midwest
Frequently Asked Questions
How big is the demand for 1,8-dinitroanthraquinone in USA in 2025?
The global demand for 1,8-dinitroanthraquinone in USA is estimated to be valued at USD 1.5 million in 2025.
What will be the size of demand for 1,8-dinitroanthraquinone in USA in 2035?
The market size for the demand for 1,8-dinitroanthraquinone in USA is projected to reach USD 2.0 million by 2035.
How much will be the demand for 1,8-dinitroanthraquinone in USA growth between 2025 and 2035?
The demand for 1,8-dinitroanthraquinone in USA is expected to grow at a 2.9% CAGR between 2025 and 2035.
What are the key product types in the demand for 1,8-dinitroanthraquinone in usa?
The key product types in demand for 1,8-dinitroanthraquinone in USA are purity ≥98%, purity ≥99% and other purity configurations.
Which application segment to contribute significant share in the demand for 1,8-dinitroanthraquinone in USA in 2025?
In terms of application, pharmaceutical manufacturing segment to command 52.7% share in the demand for 1,8-dinitroanthraquinone in USA in 2025.
link





